_______ Are Different Forms Of A Single Element.
comoprofessor
Nov 30, 2025 · 12 min read
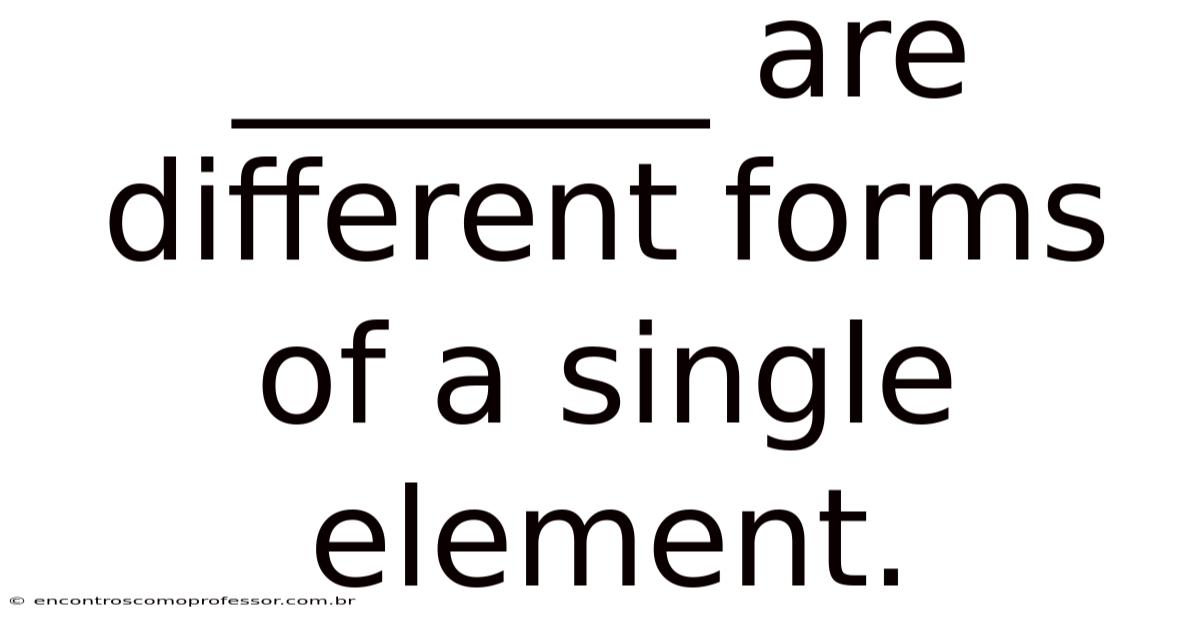
Table of Contents
Allotropes are different forms of a single element.
Imagine graphite and diamonds, both made entirely of carbon atoms. Yet, one is soft and used in pencils, while the other is renowned for its hardness and brilliance in jewelry. This striking difference arises because carbon atoms arrange themselves in different ways, creating what we call allotropes. Allotropy, from the Greek allos (other) and tropos (manner), refers to the ability of an element to exist in two or more different forms, or allotropes, in the same physical state. Each allotrope has distinct physical and chemical properties, stemming from the varied arrangements of atoms within their structures.
The concept of allotropy is fundamental in understanding the diverse behaviors of elements and their compounds. Allotropes demonstrate that the properties of matter depend not only on the type of atoms present but also on how these atoms are connected. Understanding allotropy allows scientists and engineers to tailor materials for specific applications by selecting or modifying the appropriate allotropic form. From the oxygen we breathe to the silicon in our electronics, allotropy plays a crucial role in the world around us, offering a fascinating glimpse into the versatility of the elements.
Comprehensive Overview
Allotropy is a property exhibited by certain chemical elements that can exist in two or more different forms, known as allotropes, in the same physical state. This phenomenon occurs because the atoms of the element can bond together in different ways, resulting in distinct molecular structures or crystal lattices. These structural variations lead to significant differences in physical properties such as density, hardness, color, electrical conductivity, and chemical reactivity.
The term "allotrope" was first proposed by the Swedish scientist Jöns Jacob Berzelius in 1841. He observed that certain elements could exist in different forms with varying properties. Early examples included the different forms of sulfur and carbon, which spurred further investigation into the underlying causes of allotropy. Since then, our understanding of allotropy has deepened with advancements in chemistry and physics, particularly in the fields of crystallography and quantum mechanics.
One of the primary reasons for allotropy lies in the ability of atoms to form different types of chemical bonds. This can lead to variations in molecular structure, such as different ring sizes or chain lengths, or in the arrangement of atoms in a crystal lattice. For example, carbon atoms can form strong covalent bonds in a tetrahedral arrangement, as seen in diamond, or in a hexagonal lattice, as seen in graphite. These differences in bonding result in drastically different properties; diamond is an extremely hard insulator, while graphite is a soft conductor of electricity.
Another factor contributing to allotropy is the presence of varying numbers of atoms in a molecule. Oxygen, for instance, exists as diatomic oxygen (O₂) and triatomic oxygen (O₃), also known as ozone. While O₂ is essential for respiration and combustion, O₃ is a powerful oxidant that plays a critical role in absorbing ultraviolet radiation in the Earth's atmosphere. The presence of an additional oxygen atom in ozone leads to its distinct chemical behavior and environmental impact.
Environmental conditions such as temperature and pressure can also influence the stability and prevalence of different allotropes. For example, white tin is the stable form of tin at room temperature, but at low temperatures, it transforms into gray tin, a non-metallic allotrope. This transformation, known as "tin pest," can cause the disintegration of tin objects in cold climates. Similarly, the transition between different allotropes of phosphorus is temperature-dependent, with white phosphorus being the most reactive and thermodynamically unstable form at room temperature.
The study of allotropy has significant implications in various scientific and technological fields. Understanding the properties of different allotropes allows scientists to tailor materials for specific applications. For instance, the use of graphite in lubricants and electrodes relies on its layered structure and electrical conductivity, while the use of diamond in cutting tools and abrasives is due to its extreme hardness. In the field of medicine, the allotropes of oxygen are essential for respiration and disinfection, while in electronics, the allotropes of silicon play a crucial role in semiconductor devices.
Allotropy is not limited to non-metals; several metals also exhibit this phenomenon. Iron, for example, has different allotropes that are stable at different temperatures. These allotropes have distinct crystal structures, which affect the mechanical properties of steel. The control of these allotropic transformations is crucial in the heat treatment of steel to achieve desired strength and hardness. Similarly, plutonium has multiple allotropes, each with different densities and crystal structures, which is important in nuclear applications.
The exploration of allotropy continues to drive innovation in materials science and nanotechnology. The discovery of fullerenes, carbon nanotubes, and graphene has opened up new possibilities for creating advanced materials with unique properties. These allotropes of carbon have found applications in electronics, energy storage, and composite materials. Furthermore, the study of allotropy in other elements may lead to the discovery of new materials with unprecedented properties, pushing the boundaries of scientific and technological advancements.
Trends and Latest Developments
Recent trends in the study of allotropy have focused on the synthesis and characterization of novel allotropes, as well as the exploration of their potential applications in cutting-edge technologies. Scientists are continuously seeking to create new forms of elements with enhanced or unique properties. This has led to significant advancements in materials science, nanotechnology, and related fields.
One notable trend is the increasing interest in two-dimensional (2D) materials, which are essentially single-layer allotropes of certain elements. Graphene, a 2D allotrope of carbon, has garnered immense attention due to its exceptional mechanical strength, electrical conductivity, and thermal properties. Its discovery has spurred research into other 2D materials, such as silicene (a 2D allotrope of silicon), germanene (germanium), and phosphorene (phosphorus). These materials hold promise for applications in flexible electronics, sensors, and energy storage devices.
Another significant development is the exploration of allotropes under extreme conditions, such as high pressure and temperature. These conditions can induce structural transformations and create new allotropes with unusual properties. For example, high-pressure experiments have led to the synthesis of new allotropes of carbon with hardness exceeding that of diamond. These superhard materials have potential applications in cutting tools, abrasives, and other high-performance materials.
Computational methods, such as density functional theory (DFT), are playing an increasingly important role in the study of allotropy. These methods allow scientists to predict the stability and properties of hypothetical allotropes before attempting their synthesis in the laboratory. Computational studies can also provide insights into the mechanisms of allotropic transformations and the factors that govern the stability of different allotropes.
The convergence of experimental and computational approaches is accelerating the discovery and characterization of new allotropes. Researchers are using advanced techniques such as X-ray diffraction, electron microscopy, and Raman spectroscopy to characterize the structure and properties of newly synthesized allotropes. These techniques provide valuable information about the atomic arrangement, bonding, and electronic structure of the materials.
In addition to the synthesis and characterization of new allotropes, there is growing interest in tailoring the properties of existing allotropes through chemical modification and doping. For example, the electronic properties of graphene can be tuned by introducing defects or doping it with other elements. This allows scientists to create graphene-based materials with specific functionalities for various applications.
The latest research also focuses on the environmental and health impacts of different allotropes. Some allotropes, such as asbestos (a fibrous form of silicate minerals), are known to be harmful to human health. Understanding the toxicity of different allotropes is crucial for developing safe handling and disposal procedures. Furthermore, the environmental fate and transport of allotropes, such as nanomaterials, are being investigated to assess their potential impact on ecosystems.
From a professional insight perspective, the future of allotrope research looks promising. As our understanding of the fundamental principles governing allotropy deepens, we can expect to see the discovery of more novel allotropes with unprecedented properties. These materials have the potential to revolutionize various industries, including electronics, energy, medicine, and aerospace. The ongoing research efforts in this field are likely to lead to significant technological advancements and societal benefits in the years to come.
Tips and Expert Advice
Understanding and utilizing allotropes effectively requires a blend of theoretical knowledge and practical skills. Here are some tips and expert advice to help you navigate the fascinating world of allotropes:
- Know the Basics: Before diving into advanced topics, ensure you have a solid understanding of the fundamental concepts of allotropy. This includes knowing which elements exhibit allotropy, the different types of bonding arrangements that lead to allotropes, and the factors that influence their stability. Reviewing textbooks, scientific articles, and online resources can provide a strong foundation.
- Stay Updated: The field of allotropy is rapidly evolving, with new discoveries and advancements being made regularly. Keep abreast of the latest research by reading scientific journals, attending conferences, and following experts in the field. Subscribing to relevant newsletters and online forums can also help you stay informed about emerging trends and technologies.
- Understand Applications: Different allotropes have unique properties that make them suitable for specific applications. Familiarize yourself with the various uses of allotropes in different industries, such as electronics, materials science, and medicine. For example, understanding the properties of carbon allotropes like graphene and carbon nanotubes is crucial for developing advanced electronic devices and composite materials.
- Experimental Techniques: If you are involved in experimental research, mastering the techniques used to synthesize, characterize, and manipulate allotropes is essential. This includes techniques such as chemical vapor deposition (CVD), exfoliation, and high-pressure synthesis. Additionally, learn how to use advanced characterization methods like X-ray diffraction, electron microscopy, and Raman spectroscopy to analyze the structure and properties of allotropes.
- Computational Modeling: Computational methods are increasingly used to predict the properties of new allotropes and to understand the mechanisms of allotropic transformations. Familiarize yourself with computational techniques such as density functional theory (DFT) and molecular dynamics simulations. These tools can help you design new materials with desired properties and optimize experimental conditions for their synthesis.
- Safety Precautions: Some allotropes can be hazardous to human health or the environment. For example, white phosphorus is highly reactive and toxic, while certain nanomaterials may have adverse effects if inhaled or ingested. Always follow proper safety protocols when handling allotropes, including wearing appropriate personal protective equipment (PPE) and disposing of waste materials responsibly.
- Collaborate and Network: The study of allotropy is often interdisciplinary, requiring expertise from various fields such as chemistry, physics, materials science, and engineering. Collaborate with researchers from different backgrounds to gain new perspectives and insights. Networking with experts in the field can also provide valuable opportunities for learning and career advancement.
- Real-World Examples: Consider the example of tin. At room temperature, white tin is the stable allotrope and is used in many applications, including plating steel cans to prevent corrosion. However, at temperatures below 13.2°C, white tin can transform into gray tin, a non-metallic allotrope. This transformation, known as "tin pest," can cause the disintegration of tin objects. Understanding this phenomenon is crucial for preserving historical artifacts and designing reliable electronic devices.
By following these tips and expert advice, you can enhance your understanding of allotropes and their applications, and contribute to the advancement of this exciting field.
FAQ
Q: What is allotropy? A: Allotropy is the property of some chemical elements to exist in two or more different forms, known as allotropes, in the same physical state. These allotropes have different physical and chemical properties due to variations in their atomic arrangements or molecular structures.
Q: Which elements exhibit allotropy? A: Many elements exhibit allotropy, including carbon, oxygen, sulfur, phosphorus, and tin. Each of these elements can exist in multiple allotropic forms with distinct properties.
Q: What causes allotropy? A: Allotropy is caused by the ability of atoms to bond together in different ways, leading to variations in molecular structure or crystal lattice. Factors such as temperature, pressure, and the presence of impurities can also influence the stability and prevalence of different allotropes.
Q: How do allotropes differ in properties? A: Allotropes can differ significantly in physical properties such as density, hardness, color, electrical conductivity, and chemical reactivity. For example, diamond and graphite, both allotropes of carbon, have vastly different properties due to their different bonding arrangements.
Q: Can metals exhibit allotropy? A: Yes, some metals can exhibit allotropy. Iron, for example, has different allotropes that are stable at different temperatures. These allotropes have distinct crystal structures, which affect the mechanical properties of steel.
Q: What are some applications of allotropes? A: Allotropes have numerous applications in various fields. Diamond is used in cutting tools and abrasives due to its extreme hardness, while graphite is used in lubricants and electrodes due to its layered structure and electrical conductivity. Oxygen is essential for respiration and combustion, while ozone is used for disinfection.
Q: How are new allotropes discovered? A: New allotropes can be discovered through experimental synthesis under extreme conditions, such as high pressure and temperature. Computational methods, such as density functional theory (DFT), can also be used to predict the stability and properties of hypothetical allotropes before attempting their synthesis in the laboratory.
Q: Are some allotropes harmful? A: Yes, some allotropes can be harmful to human health or the environment. For example, white phosphorus is highly reactive and toxic, while asbestos (a fibrous form of silicate minerals) is known to be carcinogenic. Proper safety precautions should be taken when handling potentially hazardous allotropes.
Conclusion
Allotropes are different forms of a single element, each exhibiting unique properties due to variations in atomic arrangement or molecular structure. From the hardness of diamond to the conductivity of graphite, allotropy plays a crucial role in shaping the properties of materials and their applications in various fields. Understanding the principles of allotropy allows scientists and engineers to tailor materials for specific purposes, leading to advancements in technology and innovation.
The study of allotropy continues to evolve with the discovery of new allotropes and the exploration of their potential applications. As we delve deeper into the world of materials science, the versatility of allotropes promises to unlock new possibilities in electronics, energy, medicine, and beyond.
To further explore this fascinating topic, consider delving into specific elements and their allotropes. Share your insights or questions in the comments below to continue the conversation and deepen our collective understanding of the diverse world of allotropes.
Latest Posts
Related Post
Thank you for visiting our website which covers about _______ Are Different Forms Of A Single Element. . We hope the information provided has been useful to you. Feel free to contact us if you have any questions or need further assistance. See you next time and don't miss to bookmark.